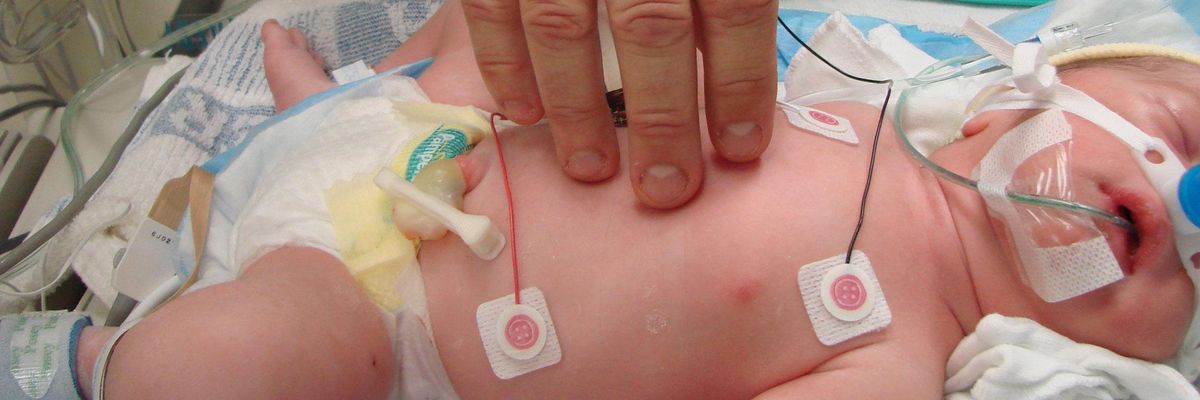
Six women in Congress are demanding federal regulators take steps to remove phthalates and other hormone-hijacking chemicals from medical products, especially IV bags and neonatal equipment.
The U.S. has made "minimal progress" in reducing harmful exposures in the United States over the past 20 years—even as alternatives become available and other jurisdictions, notably Europe and California, take steps to reduce patient risk, say Reps. Katie Porter, Jackie Speier, Anna Eshoo, Lucille Roybal-Allard (all D-Calif.), Susan Wild (D-Pa.) and Jan Schakowsky (D-Ill.).
"Patients should not be exposed to phthalates and EDCs [endocrine-disrupting chemicals] when they seek medical treatment," the representatives wrote in a letter to acting Food and Drug Administration chief Dr. Janet Woodcock. "It's also not something parents should worry about when their infant is receiving critical treatment in the neonatal intensive care unit."
The Congresswomen called on the FDA to create a "senior level" working group to do three things:
- Review and update FDA guidelines on the use of phthalates and other hormone-hijacking chemicals in IV bags and other medical equipment;
- Identify and recommend rules needed to protect patients from toxics exposure in medical products; and
- Establish an education program to build "clinician awareness" of the risks of using medical devices with toxic additives like DEHP.
"The time for action on this health risk is long overdue," the lawmakers said.
Evidence of DEHP harm to reproduction, fertility
Manufacturers add phthalates like DEHP to plastic products to increase flexibility and reduce brittleness. Some medical products such as IV bags and tubing can contain up to 40 percent DEHP by weight, according to an analysis by Health Care Without Harm.
Those additives leach to varying degrees from medical devices. DEHP is particularly problematic, as extensive research suggests exposures during critical periods of development can interfere with testosterone production and disrupt normal male reproductive development.
The European Union considers DEHP a reproductive toxicant and an endocrine disruptor; rules developed in 2017 require risk-benefit analysis before phthalates like DEHP can be used in medical devices.
California has declared DEHP a reproductive and developmental toxicant and a carcinogen and advises patients to request DEHP-free devices when seeking medical care.
Related: The danger of hormone-mimicking chemicals in medical devices and meds
Meanwhile the FDA hasn't updated its rules since 2002, when it recommended health care providers "consider" alternatives to DEHP when treating high-risk patients.
That needs to change, the lawmakers say.
"Despite these findings and the growing body of evidence that has confirmed earlier research and identified additional risks of adverse health effects on vulnerable patients, there has been minimal progress in the U.S. over the last 20 years in reducing the use of DEHP in medical devices," the lawmakers said.
"It is time for a reckoning in the healthcare community to address our role in exposing patients to potentially harmful chemicals."
Download a copy of the letter here:
Letter to FDA about DEHP IV Bags.pdf
Banner photo by Martin LaBar/flickr